Artificial cells are simplified synthetic systems designed to simulate key functions of natural cells, providing a powerful platform to study and understand biological properties1. These models can be derived from various materials, including liposomes, polymersomes, lipid/polymer hybrids, natural cell membranes, colloidosomes, metal-organic frameworks, and coacervates.
Among these, liposomes are considered the optimal artificial cell model because they possess the same phospholipid bilayer structure as natural cells and can efficiently encapsulate biomolecules1. While construction methods include thin-film hydration, electroformation, and phase transfer, microfluidic double emulsion templating has emerged as a superior approach2. This technology allows for the highly efficient and controllable generation of stable, uniform "cell templates" (W/O/W droplets), serving as a robust foundation for biomimetic membrane research and synthetic biology3.
All solutions should be filtered using a 0.2 μm syringe filter.
The setup uses a DUAL model capillary droplet microfluidic chip. It is composed of fully removable parts: a hexagonal prism-shaped glass chip body with mounting holes, coaxially-aligned capillary tubes and capillary tube adjustment assemblies.
The DUAL specific design allows for multiple liquid type emulsification within the same device.
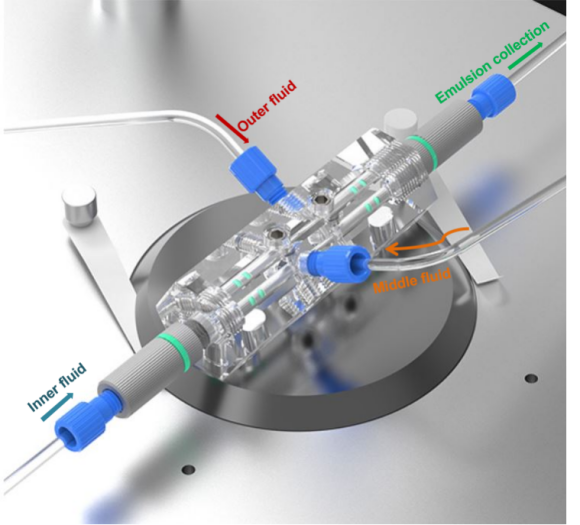
Figure 1. DUAL chip design
This workstation combines intuitive syringe pump control with visualization system. It is an integrated solution for any lab wanting to adopt droplet microfluidics technology. It is perfect for many applications: particle generation, encapsulation, emulsions... and many more!
Before assembly, the injection capillary of the DUAL chip must undergo hydrophobic treatment using octadecyltrimethoxysilane.
Load the IA, LO, and OA solutions into 10 mL syringes and fix them to the workstation's syringe pumps. Connect syringes to the chip inlets via tubing. Add an appropriate amount of collection phase to a collection bottle, ensuring the outlet tube is submerged to facilitate smooth droplet entry.
Typical flow rate ranges are set as follows:
IA: 1–10 μL/min
LO: 10–25 μL/min
OA: 80–200 μL/min
Real-time observation confirmed the successful generation of stable double emulsion droplets.
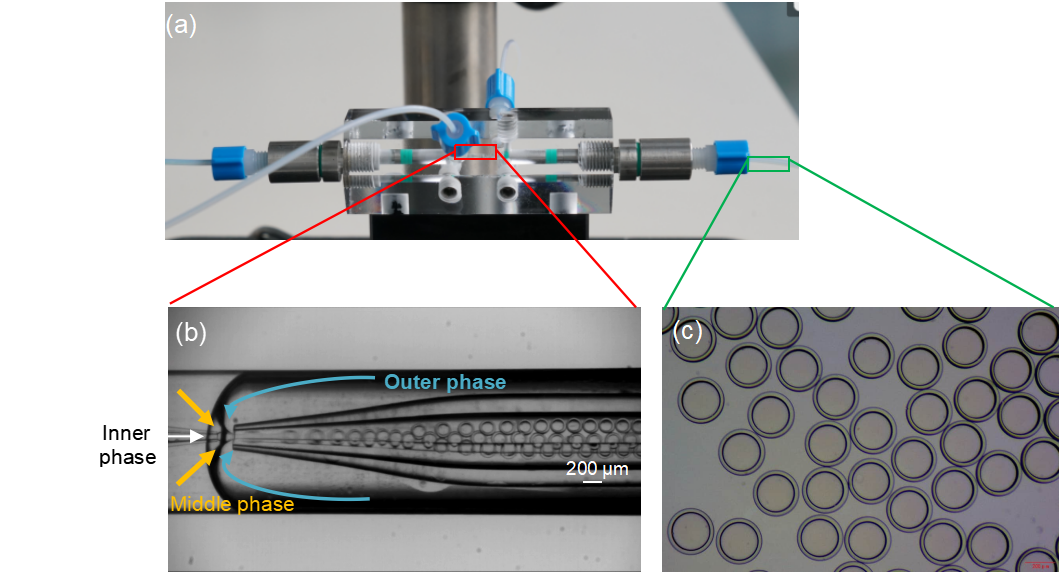
Figure 2. Double emulsion droplets formation within the DUAL chip. (a) Physical image of the DUAL chip; (b) Real-time generation of double emulsion droplets in the chip, with flow rates of the inner, middle, and outer phases being 5,15, and 90 μl/min, respectively; (c) Optical microscope image of double emulsion droplets on a glass slide.
The dimensions and stability of the artificial cell templates can be precisely tuned by varying the flow rates.
Increasing Qi (from 2 to 10 μL/min) while fixing Qm and Qo results in larger internal cores.
Table 1. Effect of inner phase (Qi) on double emulsions production
Case | Flow Rate (μL/min) | Junction image | ||
Inner Qi | Middle Qm | Outer Qo | ||
a | 2 | 15 | 90 |
|
b | 5 | 15 | 90 |
|
c | 10 | 15 | 90 |
|
In theory, adjusting the lipid phase flow rate (15 to 25 μL/min) allows for fine-tuning of the shell thickness. However, it was observed that when the lipid phase flow rate reaches 25 μL/min, a regime of inner fluid dripping and middle fluid jetting appears, leading to unstable double emulsion generation, as the image representative of Table 2, case c. Under such conditions, reducing the middle fluid flow rate is recommended. This observation also implies that stable double emulsion formation requires the three fluid flow rates to be maintained within a specific range.
Table 2. Effect of middle phase (Qm) on double emulsions production
Case | Flow Rate (μL/min) | Junction image | ||
Inner Qi | Middle Qm | Outer Qo | ||
a | 10 | 15 | 90 |
|
b | 10 | 20 | 90 |
|
c | 10 | 25 | 90 |
|
Increasing the outer phase flow rate (from 90 up to 210 μL/min) significantly impacts the overall droplet size and generation frequency.
Table 3. Effect of outer phase (Qo) on double emulsions production
Case | Flow Rate (μL/min) | Junction image | ||
Inner Qi | Middle Qm | Outer Qo | ||
a | 10 | 15 | 90 |
|
b | 10 | 15 | 120 |
|
c | 10 | 15 | 150 |
|
d | 10 | 15 | 180 |
|
e | 10 | 15 | 210 |
|
This application provides a complete solution for constructing liposome-based artificial cells using a capillary microfluidic workstation. The platform offers a robust and repeatable technical foundation, enabling researchers to achieve flexible control over shell thickness, internal compartment number, and solvent composition. This strategy facilitates the study of reversible transmembrane transport of ions, droplets, and biological macromolecules across biomimetic membranes.
1) Jiang W , Wu Z , Gao Z ,et al.Artificial Cells: Past, Present and Future. ACS Nano 2022, 16, 10, 15705–15733.
2) Lu, Y.; Allegri, G.; Huskens, J. Vesicle-Based Artificial Cells: Materials, Construction Methods and Applications. Mater. Horiz. 2022, 9, 892−907.
3) Liu J , Pan Y , Liu Z ,et al.Advancements in Artificial Cell Preparation and Biomedical Applications. Macromolecular Bioscience, 2025, 25(8):2500052.