Core-shell microcapsules are specialized structures designed to encapsulate active materials within a protective core. These capsules isolate, stabilize, and protect ingredients from the surrounding environment while serving as controlled-release systems for drugs, fragrances, catalysts, and self-healing agents across industries ranging from pharmaceuticals to textiles.
To achieve stability and structural integrity, microcapsules require effective solidification. UV-initiated polymerization (Photo-curing) is a widely adopted method due to its rapid response time—initiating polymerization within seconds—and its ability to provide precise spatial control over the curing area. While traditional methods like spray drying or phase separation struggle with size uniformity, droplet microfluidics allows for the precise manipulation of fluids to create highly monodisperse microcapsules with controllable dimensions and shell thickness. This application note demonstrates a professional solution for the production of GMA/ETPTA-based microcapsules using a UV-crosslinking microfluidic workstation.
All solutions should be filtered using a 0.2 μm syringe filter.
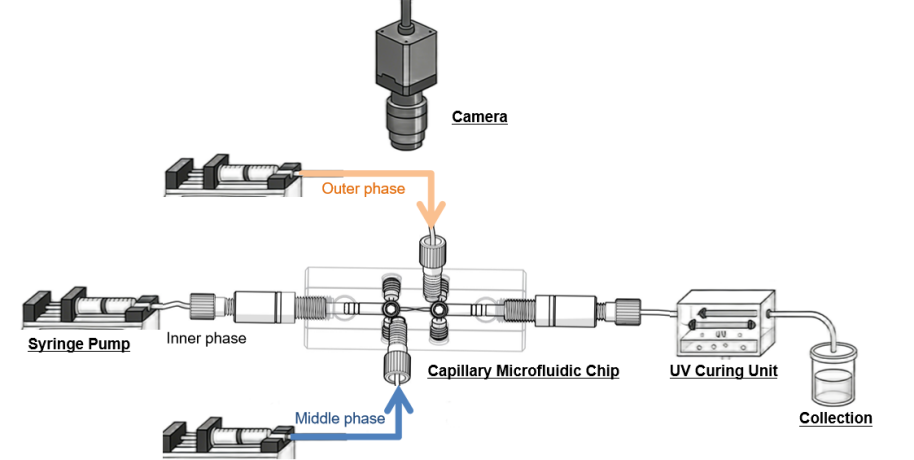
Figure 1. Experimental set-up to produce double emulsions.
The experimental setup for generating double emulsions consists of three main components:
The fluid control system is centered around the MF-3G Microfluidic Workstation. This workstation integrates 3 intuitive syringe pump control. It features a combination of three individual syringe pumps, allowing for both complex and routine experimental procedures to be performed with precision and flexibility.

Figure 2. MF-3G Microfluidic Workstation.
The setup uses a DUAL model capillary droplet microfluidic chip. It is composed of fully removable parts: a hexagonal prism-shaped glass chip body with mounting holes, coaxially-aligned capillary tubes and capillary tube adjustment assemblies.The DUAL specific design allows for multiple liquid type emulsification within the same device. Note: The injection capillary of the DUAL chip requires hydrophobic treatment before use.
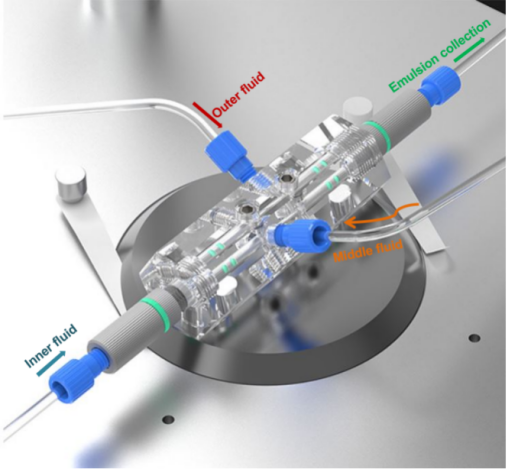
Figure 3. DUAL chip design
A dedicated UV module is incorporated into the setup, enabling photocuring or UV-triggered reactions as required for specific double emulsion formulations.
Before assembly, the injection capillary of the DUAL chip must undergo hydrophobic treatment using octadecyltrimethoxysilane.
Load the inner, middle, and outer solutions into 10 mL syringes and secure them to the workstation's syringe pumps.
Connect the syringes to the DUAL chip inlets using PTFE tubing and Luer-to-inverted-cone connectors to ensure a leak-proof system.
Set the flow rates on the workstation. The recommended sequence is:
Start the Outer Phase first to prime the channels.
Once stable, start the Middle Phase to form a single emulsion.
Finally, start the Inner Phase to generate the W/O/W double emulsion.
The generated double emulsions flow from the chip outlet and are subjected to UV light (365 nm) at a distance of 1 cm to trigger cross-linking and solidify the shell.
Real-time imaging confirms the generation of monodisperse double emulsions within the DUAL chip. The inclusion of Trypan Blue in the inner core provides a sharp visual distinction between the aqueous core and the GMA/ETPTA shell.
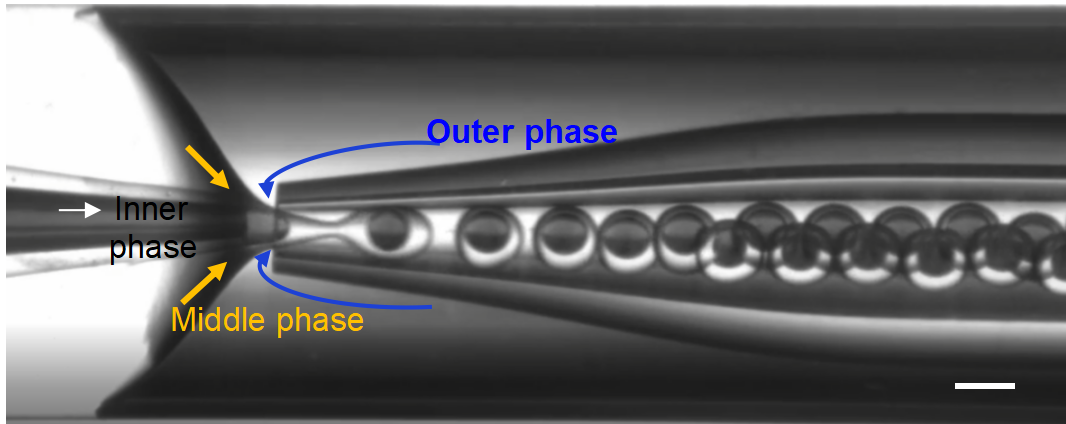
Figure 4. Real-time generation of double emulsion droplets in the DUAL chip, with flow rates of the inner, middle, and outer phases being 12, 20, and 50 μl/min, respectively. Scale bar: 200 μm.

Figure 5. Optical microscope images of double emulsion obtained at the output(a) and UV-crosslinked microcapsules(b).
Before UV treatment, the droplets exist as fluid double emulsions. Upon exposure to high-intensity UV light, the acrylate-based shell undergoes rapid cross-linking, resulting in mechanically stable solid microcapsules. These capsules maintain their spherical integrity and monodispersity after the curing process. Real-time observation confirmed the successful generation of stable double emulsion droplets.
The MF-3G Microfluidic Workstation, when integrated with a UV module and the droplet generation chip (DUAL model Glass Capillary Based Microfluid ic Chip), provides a highly effective platform for the production of UV-crosslinked microcapsules.
This platform opens the door to numerous advanced applications, including:
1) Mettler M , Dewandre A , Tumanov N ,et al. Single crystal formation in core–shell capsules. Chemical Communications, 2023, 59(85):4.
2) Hamonangan W M, Lee S, Choi Y H, et al. Osmosis-mediated microfluidic production of submillimeter-sized capsules with an ultrathin shell for cosmetic applications. ACS Applied Materials & Interfaces, 2022, 14(16): 18159-18169.
3) Liu H, Piper J A, Li M. Rapid, simple, and inexpensive spatial patterning of wettability in microfluidic devices for double emulsion generation. Analytical Chemistry, 2021, 93(31): 10955-10965.
4) Zhao Y, Moshtaghibana S, Zhu T, et al. Microfluidic fabrication of novel polymeric core‐shell microcapsules for storage of CO2 solvents and organic chelating agents. Journal of Polymer Science, 2022, 60(11): 1727-1740.
5) Lee S, Lee T Y, Kim D J, et al. Osmotic-stress-mediated control of membrane permeability of polymeric microcapsules. Chemistry of Materials, 2018, 30(20): 7211-7220.
6) Loiseau E, Rühs P A, Hauser A, et al. Strong Dual-Compartment Microcapsules Loaded with High Cargo Contents. Langmuir, 2018, 34(1): 205-212.
7) Kim B, Jeon T Y, Oh Y K, et al. Microfluidic production of semipermeable microcapsules by polymerization-induced phase separation. Langmuir, 2015, 31(22): 6027-6034.
8) Elsafi A M, Bahrami M. A novel spherical micro-absorber for dehumidification systems. International Journal of Refrigeration, 2024, 157: 73-85.