P4HB stands out among polyhydroxyalkanoates (PHAs) as the only material with FDA clearance for clinical usage since 2007. It possesses a linear chain structure composed of 4-
hydroxybutyrate monomers and exhibits mechanical properties comparable to ultra-high molecular weight polyethylene, with an elongation at break of around 1000%.
Compared to polylactic acid (PLA), P4HB is more flexible, has a slower degradation rate, and its degradation product (4-hydroxybutyrate) is a natural metabolite in the human body with lower acidity, leading to minimal inflammatory response. Droplet-based microfluidics offers a precise alternative for generating monodisperse P4HB microspheres, which are essential for applications requiring consistent drug release profiles or uniform scaffolding in bone tissue repair.
• Polymer: P4HB (Mw = 1, 114, 000).
• Surfactant: Poly(vinyl alcohol) (PVA, Mw = 67, 000).
• Solvent: Dichloromethane (DCM) .
This workstation combines intuitive syringe pump control with visualization system. It is an integrated solution for any lab wanting to adopt droplet microfluidics technology.
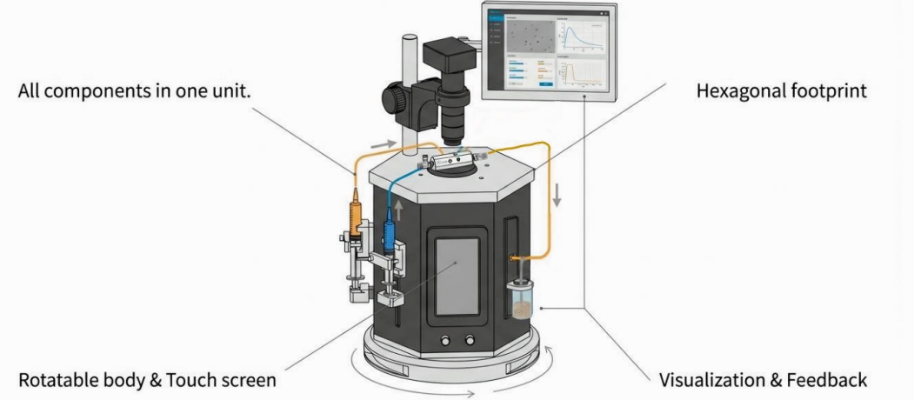
Figure 1. Scheme of the setup for P4HB microdroplets production

Figure 2. MONO chip design
The MONO chip is an assembled glass capillary microfluidic device for single droplet generation. It is composed of fully removable parts: a hexagonal prism-shaped glass chip body with mounting holes, coaxially-aligned capillary tubes and capillary tube adjustment assemblies.
The MONO specific design allows for single emulsions fabrication.
Dispersed Phase (DP): Dissolve P4HB in DCM at room temperature to a concentration of 5% (w/w).
Continuous Phase (CP): Prepare a 2% (w/v) PVA aqueous solution.
Ensure all solutions are filtered to avoid clogging the capillaries.
Connect syringes containing the dispersed phase (DP) and continuous phase (CP) to the chip inlets via tubing.
Start CP first at a low rate to wet channels. Then introduce DP. Continue both flows until all air bubbles are purged and steady flow is achieved.
Adjust the flow rate ratio (CP:DP) to tune droplet size: higher flow rate ratio = Smaller droplets.
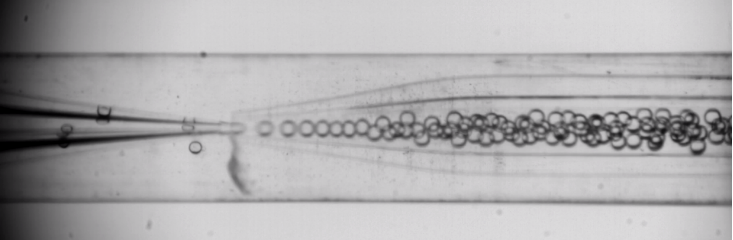
Figure 3. P4HB droplet formation with nozzle of 60µm in injection capillary
and of 140µm in collection capillary
The microdroplets are collected into 2% PVA bath and then placed at room temperature to allow the DCM evaporation, the P4HB polymer chains spontaneously rearrange and solidify, forming solid microspheres.
After solidification, the microspheres are washed via multiple centrifugation cycles (typically >10) with high-purity water until the supernatant is clear, effectively removing residual surfactants and water-soluble impurities.
Finally, lyophilize the cleaned dispersion to obtain white powder of P4HB microparticles for long-term storage.
The microparticle size and monodispersity are determined under a microscope. To do so, collect a small sample by placing the outlet tubing directly on a glass cover slip. The microfluidic system demonstrated exceptional control over particle size. Table 1 details the dependence of particle size on the flow rate ratio, showing the size variation immediately after droplet formation and the final diameter after several minutes.
Table 1: Influence of flow rate ratios on size
DP | CP | Droplet Diameter | Piture | Microparticle Diameter | Piture |
10 µL/min | 160 µL/min | 152 µm |
| 60µm |
|
10 µL/min | 170 µL/min | 140 µm |
| 51µm |
|
10 µL/min | 200 µL/min | 90 µm |
| 36µm |
|
A microparticle size population has been analyzed to determine microparticles monodispersity at 3%. The monodispersity has been analyzed on a microparticle population (around 200 microparticles). The flow rates have been adapted to generate 50 µm diameter particle size.
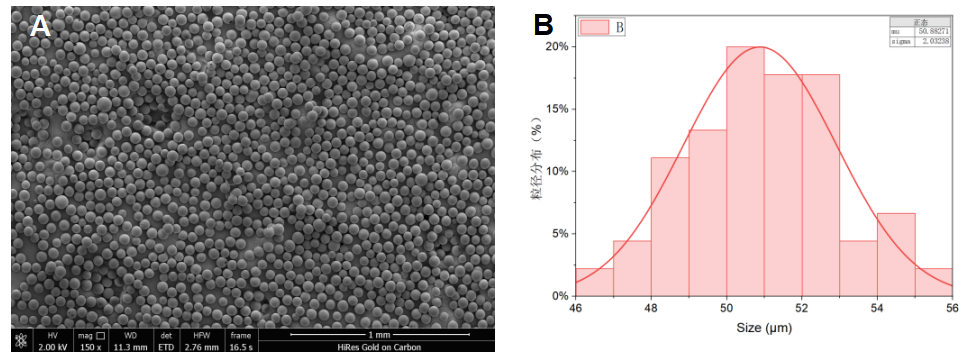
Figure 4. Particle size distribution: A) Scanning electron microscope (SEM) images of P4HB microparticles B) Particle monodispersity graph
By utilizing the MF-2G workstation and a MONO chip, we successfully fabricated monodisperse P4HB microparticles. This platform offers a robust and scalable method for producing high-strength, flexible biomaterials that meet the stringent requirements of clinical and tissue engineering applications.
1) Utsunomia C, Ren Q and Zinn M (2020) Poly(4-Hydroxybutyrate): Current State and Perspectives. Front. Bioeng. Biotechnol. 8:257. doi: 10.3389/fbioe.2020.00257