In recent years, biodegradable microspheres have gained widespread importance in the delivery of bioactive agents. Poly(lactic-co-glycolic acid) (PLGA) is one of the most successful materials due to its excellent biocompatibility and FDA-approved safety profile. While PLGA microparticle synthesis is successful, traditional tools often suffer from wide size distribution and poor repeatability.
To solve these problems, droplet-based microluidics offers an efficient method for improvement. This application note describes a method to synthesize porous PLGA microspheres by combining gas-foaming (via porogen decomposition) and microfluidic precision, providing a controllable way to tune porosity and particle size.
• Polymer: PLGA (82/ 18 L-lactide/glycolide).
• Surfactant: Poly(vinyl alcohol) (PVA) .
• Porogen: Ammonium bicarbonate ( NH4HCO3 ) .
• Solvent: Dichloromethane (DCM) .
• Hydrolysis Agent: Sodium hydroxide ( NaOH) .
The MONO chip is an assembled glass capillary microfluidic device for single droplet generation. It is composed of fully removable parts: a hexagonal prism-shaped glass chip body with mounting holes, coaxially-aligned capillary tubes and capillary tube adjustment assemblies.

Figure 1. MONO chip design
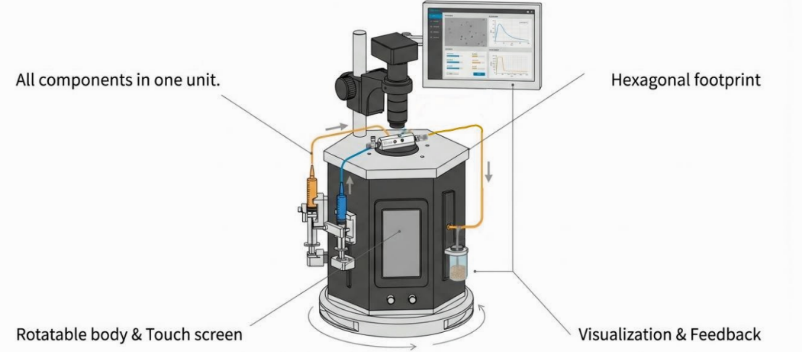
Figure 2. Porous PLGA microparticle production station
This workstation combines intuitive syringe pump control with visualization system. It is an integrated solution for any lab wanting to adopt droplet microfluidics technology. It is perfect for many applications: particle generation, encapsulation, emulsions... and many more!
Dissolve PLGA in DCM to obtain a 2 wt% PLGA solution. Add 2 mL of 1 % ( w / v ) NH4HCO3 aqueous solution to 20 mL of this PLGA solution. The mixture was sonicated using an ultrasonic homogenizer at 60% power for 1 minute to form the primary emulsion (W1/O).
Connect syringes containing the primary emulsion (dispersed phase) and continuous phase (0.5% (w/v) PVA solution) to the chip inlets via tubing.
Start continous phase first at a low rate to wet channels. Then introduce dispersed phase. Continue both flows until all air bubbles are purged and steady flow is achieved.
Adjust the flow rate ratio (continous phase:dispersed phase) to tune droplet size:, Normally, higher flow rate ratio = Smaller droplets.
The double emulsion (W1/O/W2 ) microdroplets are collected into a reservoir filled with the continous phase and then placed at room temperature to allow the DCM evaporation.
After solidification, the microspheres are washed via multiple centrifugation cycles (typically >10) with high-purity water until the supernatant is clear, effectively removing residual surfactants and water-soluble impurities.
To control porosity, immerse the washed microspheres (MPs)in 0.2M NaOH solution. This alkaline treatment facilitates the exposure of the porous scaffold. Thoroughly wash three times with water to remove NaOH residues.
Finally, the solution of washed particles was frozen overnight in -80℃ and lyophilized to produce a dry powder of particles that was stored at -20℃.
Once droplets are generated, the solvent inside the microparticles in the presence of the continuous phase will continuously diffuse out of the droplet. With solvent diffusion outside of the droplet the PLGA concentration will increase and precipitate to form a solid microparticle smaller than the droplet.
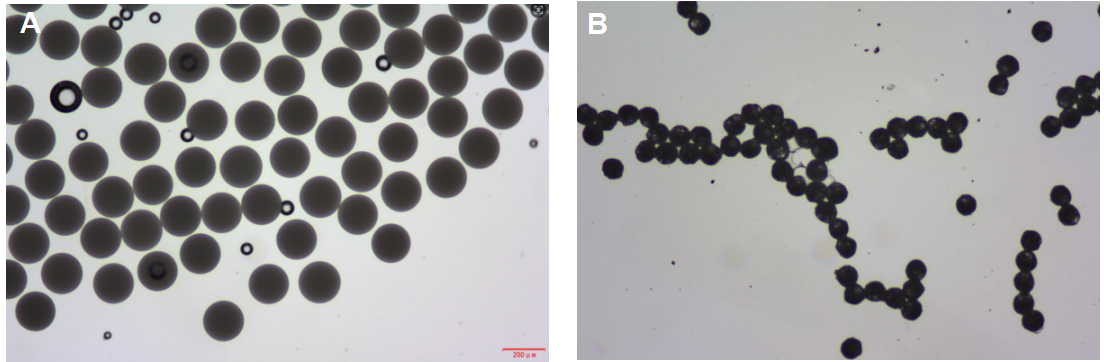
Figure 3: Particle shrinking during solvent evaporation: A) Particle size just after
droplet formation; B) Particle size after few minutes.
The NaOH hydrolysis treatment successfully yielded PLGA microspheres with a distinct porous structure. The monodispersity was significantly improved compared to traditional batch methods.
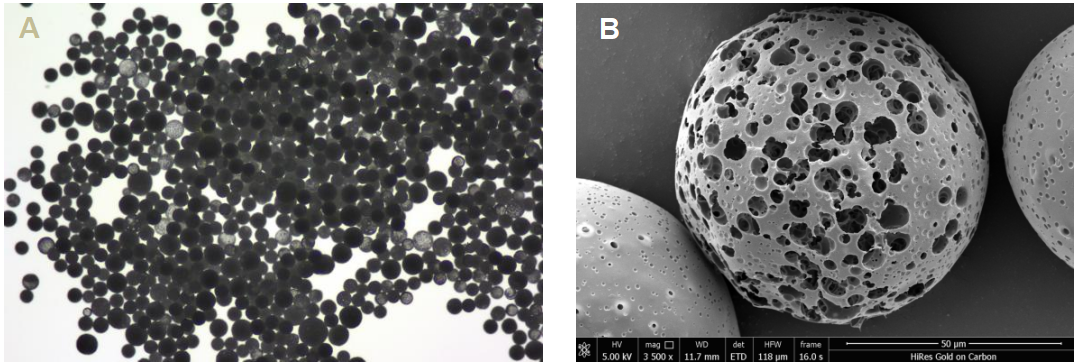
Figure 4: A) Microscope observation of PLGA porous microsphere particles just after the lyophilization; B) Scanning electron microscope (SEM) images of PLGA porous microsphere particles.
This study demonstrates the successful fabrication of porous PLGA microspheres using a glass capillary coaxial chip platform. The integration of microluidic precision and post-hydrolysis treatment allows for the production of particles with a controllable hollow porous structure.
1) Amoyav, B., Benny, O. Microluidic Based Fabrication and Characterization of Highly Porous Polymeric Microspheres. Polymers 2019 , 11 , 419.
2) Su, Y. et al. PLGA-Based Biodegradable Microspheres in Drug Delivery: Recent Advances in Research and Application. Drug Delivery 2021, 28 (1), 1397–1418.