Introduction to Emulsions and Droplets
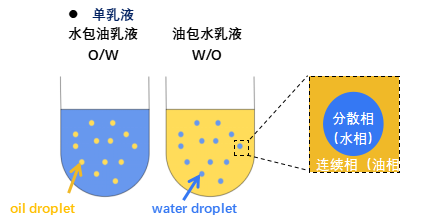
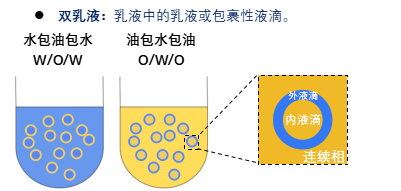
An emulsion is a mixed system of two or more immiscible liquids, in which one phase is dispersed in the other in the form of droplets. The phase where the droplets exist is called the dispersed phase or internal phase, and the other phase is called the continuous phase or external phase. Emulsions are generally divided into single emulsions and double emulsions. The most common single emulsions are oil-in-water (O/W) and water-in-oil (W/O) types. In an O/W emulsion, the continuous phase is water and the dispersed phase is oil, while in a W/O emulsion, the continuous phase is oil and the dispersed phase is water. Compared with single emulsions, double emulsions are more complex systems, also known as emulsions within emulsions, which have two main types: water-in-oil-in-water (W/O/W) and oil-in-water-in-oil (O/W/O).
Emulsions have the following multiple advantages and are widely used in food, pharmaceutical, and cosmetic industries[1]:
• Small emulsion droplet size, large specific surface area, and high mass and heat transfer efficiency;
• Ability to carry out various biological and chemical reactions inside with minimal sample and reagent consumption;
• Closed reaction system that avoids cross-contamination of samples;
• Capability to isolate, protect, and achieve controlled release of substances such as drugs and active ingredients.
ntroduction to Microspheres, Microgels and Microcapsules
Different from emulsion droplets, microspheres are spherical particles with diameters typically ranging from nanoscale to microscale.

Microspheres | Microgels | Microcapsules
Microspheres crosslinked via chemical bonds or physical interactions, which swell into a gel state by absorbing solvent in a good solvent, are referred to as microgels. Microcapsules are microspheres with other functional substances embedded inside. These microparticles in different states share common characteristics, such as a wide particle size range, large specific surface area, a broad selection of carrier material systems, excellent biocompatibility or biodegradability. Therefore, they are widely used in biomedical fields[2], such as drug delivery, controlled drug release, and tissue engineering.
Introduction to Droplet Microfluidics
As an important branch of microfluidic chip research, droplet microfluidics has been developed in recent years based on traditional continuous-flow microfluidic systems. It is a technology that generates and manipulates monodisperse droplets through multiphase fluid shear in microscale channels, and the formation of droplets is similar to the emulsification phenomenon mentioned above.This technology enables the controllable production of single and multiple emulsion droplets with highly monodisperse sizes, as well as the precise regulation of emulsion droplet structure (including the composition, number, size, and proportion of the internal encapsulated droplets, as well as the composition and thickness of the outer liquid layer, etc.). These droplets with precisely controllable and diverse structures can be converted into functional microspheres and core-shell microcapsule materials through subsequent methods such as polymerization, crosslinking, solvent evaporation, curing, and assembly, either inside the droplets or at the phase interface.
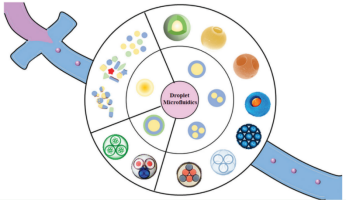
Schematic overview of the synthesis of various functional microspheres based on droplet microfluidics[3]
Why Choose Droplet Microfluidics?
Before the popularization of microfluidic methods, traditional methods for preparing various microspheres included spray drying, suspension polymerization, and emulsion evaporation. These methods have inherent drawbacks, such as unstable external force and uneven shear force during the mixing of different phases. These drawbacks result in microspheres with non-uniform and uncontrollable particle size distribution, with significant size differences ranging from tens of microns to hundreds of microns. They may even cause the loss of internal active ingredients and alter the morphology of the microspheres. In contrast, droplet microfluidics technology has the advantages of simple operation, and the prepared microspheres feature uniform size, a closed system, and excellent monodispersity. The particle size deviation can be stably controlled below 5%, and even reach 1% under specific conditions. In addition, it has the benefits of low reagent consumption, high experimental safety, controllable internal component content, and the ability to achieve more ordered internal structures.
Applications of Droplet Microfluidics
Cell-laden Matrix
Single-cell research: Droplet microfluidic systems can generate thousands of samples (droplets) per second, serving as incubators for single cells[4]. Compared with traditional cell culture in microtiter plates, microdroplets with volumes ranging from microliters (μL) to picoliters (pL) can significantly reduce the consumption of reagents and cells. Droplet-based microfluidic systems provide an analytical platform capable of isolating single cells or cell populations in individual droplets[5].
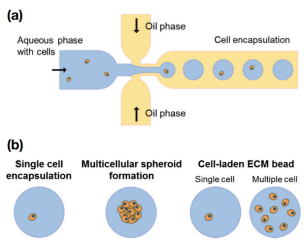
Microdroplet-based cell culture platform[6]
Tissue engineering: Droplet microfluidics technology enables the high-throughput preparation of monodisperse droplets with controllable size, composition, and function. These droplets can encapsulate one or more types of cells, and serve as templates to prepare cell-laden hydrogel microspheres with specific physicochemical properties and shear stress resistance. These microspheres can be used as scaffolds for tissue engineering, and are applied in research fields such as organ models, tumor models, and stem cell-laden injectable hydrogel scaffolds[6].
Active Substance Delivery System
Droplet microfluidics technology provides an excellent tool for the control of monodisperse particle size and desired morphology, and shows significant advantages in terms of particle structure uniformity, batch-to-batch repeatability, and drug encapsulation efficiency[7]. Microcapsules, nanoparticles, and emulsions prepared based on droplet microfluidics technology can be used as various delivery systems[8].
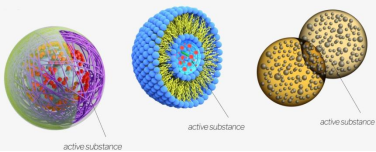
For example, various emulsions can be prepared using water/oil systems, allowing the encapsulation of multiple ingredients such as drugs, vitamins, antioxidants, fragrances, and flame retardants in the system. Through post-processing, the emulsions can be converted into hydrogel particles or core-shell microcapsules for the delivery of the above beneficial ingredients, which are widely used in pharmaceutical preparations, food, cosmetics, textiles and other industries. In addition, a nanoscale drug delivery system can be formed by encapsulating or adsorbing drugs such as active small molecule compounds, protein macromolecules, vaccines, immunotherapeutics and genes into functionalized nanocarriers (liposomes, polymer nanoparticles).
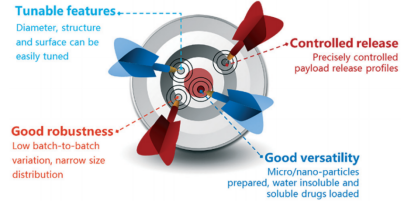
Characteristic advantages of droplet microfluidics in drug delivery[8]
Artificial Cell Synthesis
Droplet microfluidics is not limited to the synthesis of particles and microcapsules, but can also be used as a template for artificial cells. Depending on different research purposes, they can simulate specific cell functions through membrane-free compartmentalized biochemical reactions, or serve as templates for lipid bilayers. More complex designs allow the encapsulation of droplets within larger droplets to mimic the composition of real cells[9].
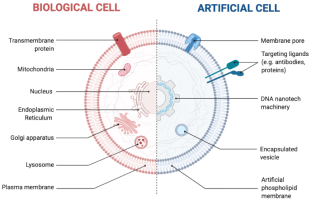
Schematic diagram of artificial cell structure
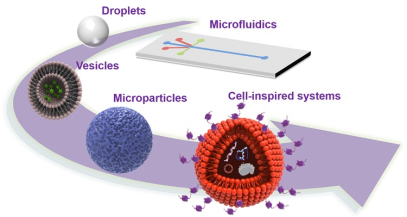
Construction of artificial cells from droplets, vesicles, polymer microspheres to cell-like systems based on microfluidic technology[9]
Biological Macromolecule Characterization
Using a water-in-oil system, droplet PCR[10] follows the workflow of component assembly, droplet generation, droplet merging, thermal cycling, and result processing, just like conventional PCR. It enables ultra-sensitive detection of specific nucleic acid sequences. Droplet-based devices have also been used to study the conditions required for protein crystallization[11] and for DNA sequencing[12].
Functional Material Synthesis & Chemical Synthesis
As a chemical synthesis method, droplets in microfluidic devices can act as individual reaction chambers. A variety of functional micro-nanoparticles, such as liquid crystal microcapsules[13], nanocrystals, and photonic crystal clusters[14], can be synthesized with the assistance of droplet microfluidics.
References
1. Lee TY, Choi TM, Shim TS, Frijns RA, Kim SH. Microfluidic production of multiple emulsions and functional microcapsules. Lab on a Chip. 2016, 16 (18): 3415–40.
2.Li, et al. Microfluidic fabrication of microparticles for biomedical applications. Chem. Soc. Rev., 2018, 47, 5646.
3.F. Long, Y. Guo, Z. Zhang, Y. Ren et al. Recent Progress of Droplet Microfluidic Emulsification Based Synthesis of Functional Microparticles. Global Challenges, 2023, 7, 2300063.
4.Joensson HN, Andersson Svahn H. Droplet microfluidics--a tool for single-cell analysis. Angewandte Chemie. 2012, 51 (49): 12176–92.
5.Jang M, Yang S, Kim P. Microdroplet-based cell culture models and their application. BioChip Journal, 2016, 10 (4): 310–317.
6.Feng Q, et al. Microgel assembly: fabrication, characteristics and application in tissue engineering and regenerative medicine. Bioactive Materials, 2021, 9: 105-119.
7.Hirvonen, et al. Microfluidic-assisted fabrication of carriers for controlled drug delivery. Lab on a chip, 2017, 17, 1856–1883.
8.Lengyel, M., Kállai-Szabó, N., Antal, V., Laki, A. J. & Antal, I. Microparticles, microspheres, and microcapsules for advanced drug delivery. Scientia Pharmaceutica, 2019, 87(3), 20.
9.Tan S, Ai Y, Yin X, et al. Recent Advances in Microfluidic Technologies for the Construction of Artificial Cells. Advanced Functional Materials, 2023, 33(45): 2305071.
10.Zheng B, Tice JD, Roach LS, Ismagilov RF. A droplet-based, composite PDMS/glass capillary microfluidic system for evaluating protein crystallization conditions by microbatch and vapor-diffusion methods with on-chip X-ray diffraction. Angewandte Chemie. 2004, 43 (19): 2508.
11.Hindson BJ, et al. High-throughput droplet digital PCR system for absolute quantitation of DNA copy number. Analytical Chemistry. 2011, 83 (22): 8604–10.
12.R. A. Adam and T. Hung. DNA sequence analysis with droplet-based microfluidics. Lab Chip, 2013, 13 , 4864-4869.
13.Chen H Q , Wang X Y , Bisoyi H K ,et al. Liquid Crystals in Curved Confined Geometries: Microfluidics Bring New Capabilities for Photonic Applications and Beyond. Langmuir. 2021, 37 (13): 3789–3807.
14.Wang JT, Wang J, Han JJ. Fabrication of advanced particles and particle-based materials assisted by droplet-based microfluidics. Small. 2011, 7 (13): 1728–54.