Droplet microfluidics technology enables the preparation of single emulsion droplets and multiple emulsion droplets with highly monodisperse sizes, as well as the precise regulation of the emulsion droplet structure (including the composition, number, size, and proportion of the internal encapsulated droplets, as well as the composition and thickness of the outer liquid layer, etc.).
These emulsion droplets with a stable and closed liquid-liquid phase interface, including water-in-oil (W/O) or oil-in-water (O/W) single emulsions, W/O/W or O/W/O double emulsions, and more complex multiple emulsions, can be used as template droplets. Microspheres or microcapsules can be obtained from these template droplets through subsequent curing methods such as polymerization, crosslinking, solvent evaporation, and assembly, either inside the droplets or at the phase interface.
Common curing methods include:
• Solvent Evaporation Method: The solvent in the droplets is evaporated to cure the dispersed phase in the droplets into microspheres. This method is suitable for the preparation of a wide range of polymer microspheres, such as polylactic acid (PLA) and poly(lactic-co-glycolic acid) (PLGA), etc.[1,2]
• Polymerization Method: This is the most commonly used method for converting emulsion droplets into solid microspheres, due to its wide range of experimental protocols and the diversity of selectable polymer materials. By adding monomers or oligomers, along with a small amount of suitable initiators (such as thermal initiators or photoinitiators) to the dispersed phase, the emulsion droplets can be polymerized under heating or ultraviolet (UV) light irradiation.
① Thermal-initiated polymerization is generally not preferred, as high temperatures may compromise the activity of biomolecules in the system, and may also cause instability of the emulsion interface, leading to coalescence of droplets before curing.
② Photoinitiated polymerization is one of the most widely used methods, as it has a fast response time and can initiate polymerization within seconds. For example, hydrogel microspheres of polyacrylamide (PAAm)[3], polyethylene glycol diacrylate (PEGDA)[4], and gelatin methacryloyl (GelMA)[5] can be obtained using W/O template droplets containing water-soluble monomers. In addition, organic polymer resin microspheres/microcapsules, such as ethoxylated trimethylolpropane triacrylate (ETPTA)[6] and tripropylene glycol diacrylate (TPGDA)[7], can be prepared using O/W oil droplets as templates.
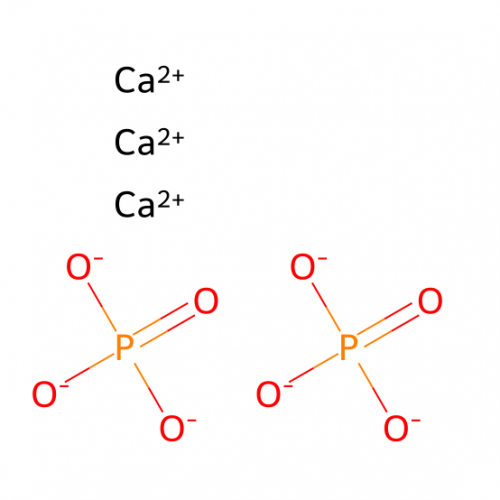
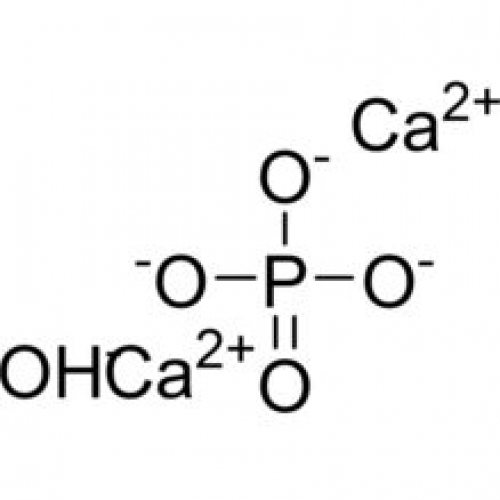
• Physical Crosslinking (Ionic Crosslinking): One of the most widely studied ionic crosslinking materials in droplet microfluidics technology is alginate hydrogel microspheres, where the gel precursor droplets are physically crosslinked by divalent cations (such as calcium ions Ca²⁺).
• Temperature-Induced Gelation and Low-Temperature Curing: The gelation method is suitable for droplets containing natural macromolecular materials, including collagen, agarose, and gelatin, which can be converted into hydrogels by simply changing the temperature. The low-temperature curing method is suitable for certain alkane and lipid materials (including glycerides) with a melting point higher than room temperature[8,9].
Aqueous Two-Phase Systems (ATPs): Droplet formation in microfluidic devices usually involves the use of an oil phase and an aqueous phase. However, droplets can also be generated in Aqueous Two-Phase Systems (ATPs), which consist of two immiscible aqueous solutions. The principle is based on the immiscibility of two hydrophilic polymers at specific concentrations. These polymers are typically polyethylene glycol (PEG) and dextran[10], which are each dissolved in water to form two distinct aqueous phases. When the concentrations of these two polymers exceed a specific critical value, phase separation occurs in the system, forming two phases that are both rich in water.
References:
[1] Q. Xu, M. Hashimoto, T. T. Dang, T. Hoare, D. S. Kohane,G. M. Whitesides, R. Langer and D. G. Anderson, Small, 2009, 5, 1575–1581.
[2] T. Watanabe, T. Ono and Y. Kimura, Soft Matter, 2011, 7, 9894–9897.
[3] S. Guo, T. Yao, X. Ji, C. Zeng, C. Wang and L. Zhang, Angew. Chem., Int. Ed., 2014, 53, 7504–7509.
[4] H. Liu, X. Qian, Z. Wu, R. Yang, S. Sun and H. Ma, J. Mater. Chem. B, 2016, 4, 482–488.
[5] C. Cha, J. Oh, K. Kim, Y. Qiu, M. Joh, S. R. Shin, X. Wang, G. Camci-Unal, K.-t. Wan, R. Liao and A. Khademhosseini, Biomacromolecules, 2014, 15, 283–290.
[6] H. Gu, B. Ye, H. Ding, C. Liu, Y. Zhao and Z. Gu, J. Mater. Chem. C, 2015, 3, 6607–6612.
[7] Visaveliya, Nikunjkumar; Köhler, J. Michael. J. Mater. Chem. C, 2015, 3(4), 844–853.
[8] Y. Zhao, H. C. Shum, L. L. A. Adams, B. Sun, C. Holtze, Z. Gu and D. A. Weitz, Langmuir, 2011, 27, 13988–13991.
[9] T. A. Comunian, A. Abbaspourrad, C. S. Favaro-Trindade and D. A. Weitz, Food Chem., 2014, 152, 271–275.
[10] L. Y. Zhang, L. H. Cai, P. S. Lienemann, T. Rossow, I. Polenz, Q. Vallmajo-Martin, M. Ehrbar, H. Na, D. J. Mooney and D. A. Weitz, Angew. Chem., Int. Ed., 2016, 55, 13470–13474.
Previous: Why does droplet microfluidics hold great potential in cosmetic R&D?
Next: Not available